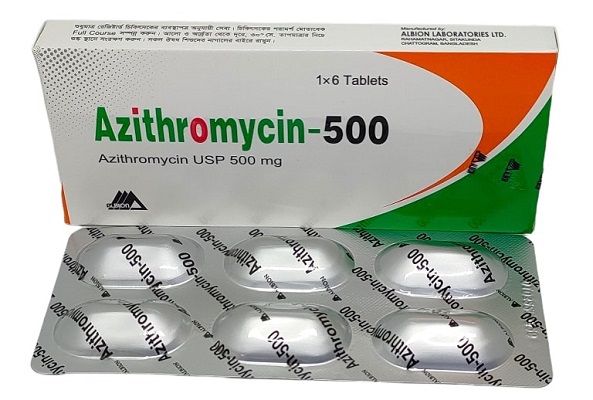
Harare, 13 April 2026 – The Medicines Control Authority of Zimbabwe (MCAZ) has urgently called for the recall of Azithromycin 500mg USP Tablets, Batch Number 24050191, after a quality defect was detected during stability testing.
This has raised concerns over the medicine’s effectiveness.
This recall follows a voluntary notification from the manufacturer after an out-of-specification (OOS) result was identified during dissolution testing conducted as part of a 24-month stability study.
Azithromycin 500mg USP Tablets, Batch Number: 24050191, is manufactured by Indoco Remedies Limited, India.
In a statement, MCAZ Director General, Richard Rukwata confirmed the development.
“MCAZ wishes to inform healthcare professionals, licensed pharmaceutical stakeholders, and the general public of a Class II recall up.
“The identified quality defect indicates non-compliance with approved product specifications, which may compromise the effectiveness of the medicine in treating bacterial infections and contribute to the development of antimicrobial resistance if therapeutic levels are not achieved.
“The Authority would like to draw the attention of all licensed wholesalers, pharmacies, public and private clinics, and hospitals that they should quarantine any of the affected units of the reported product and cooperate with Indoco Remedies Limited and local distributors of the product in Zimbabwe during the recall process.
“The general public who might have purchased the same reported batch are advised to return the product to the pharmacy where they obtained the medicine.
“Patients who recently used this product are advised to see their healthcare professionals for assistance.”
The Authority remains committed to safeguarding public health by ensuring that all medicines available in Zimbabwe meet the required standards of quality, safety, and efficacy.
MCAZ will continue to monitor the recall process and provide updates where necessary.
Medicines Control Authority of Zimbabwe (MCAZ) is a statutory body established by an Act of Parliament, the Medicines and Allied Substances Control Act (MASCA) [Chapter 15.03]. MCAZ is a successor of the Drugs Control Council (DCC) and the Zimbabwe Regional Drug Control Laboratory (ZRDCL).
DCC was established by an Act of Parliament in 1969: Drugs and Allied Substances Control Act [Chapter 15.03], following which ZRDCL became operational in 1989.
MCAZ is responsible for protecting public and animal health by ensuring that accessible medicines, allied substances, and medical devices are safe, effective, and of good quality through enforcement and adherence to standards by manufacturers and distributors.
MCAZ is a World Health Organization (WHO) Global Benchmarking Tool (GBT) Maturity Level 3 national medicines regulatory Authority, demonstrating that its regulatory systems and functions operate at a stable, well-established level in line with international standards.
The Authority applies robust regulatory processes that include product registration, licensing, Good Manufacturing Practice (GMP) inspections, laboratory testing, post-market surveillance, and pharmacovigilance.